More than 2.5 million people in Kentucky, Ohio and West Virginia have been vaccinated with at least one dose of the Moderna or Pfzier COVID-19 vaccines. Little by little, states have received more vaccine doses, but the national supply remains low.
However, more promise is on the horizon as the U.S. Food and Drug Administration approved emergency authorization use of the Johnson & Johnson vaccine. That one-dose shot is the third coronavirus vaccine available in the country, and would decrease the number of appointments necessary to be inoculated while upping overall supply.
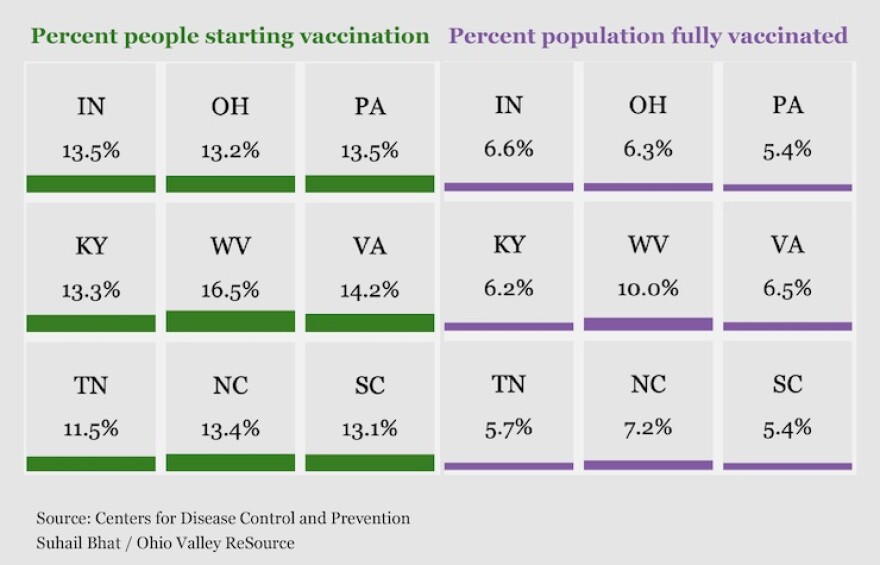
As more people become eligible for the vaccine, more questions arise about efficacy and state distribution of the vaccine. The Ohio Valley ReSource created tools to track the progress of vaccinations in Kentucky, Ohio and West Virginia, and we are asking vaccine experts to address questions from our readers and listeners.
Vince Venditto is an assistant professor in the College of Pharmacy at the University of Kentucky and specializes in vaccine design and development. He answered questions about the Pfizer and Moderna vaccines. Dr. Richard Greenberg works in the division of infectious diseases at the University of Kentucky and is the principal investigator for the Johnson & Johnson one-dose vaccine trial at the university.
Here are their answers to some of your questions and some additional resources for more information.
Variants, Infection & Efficacy
Q: What do we know about those who already had the virus taking the vaccine?
Two new studies suggest that people who’ve had COVID-19 may need only one dose of the vaccine. The studies show that one shot coupled with antibodies from the virus offers enough protection.
If someone has been infected, Venditto said, it’s recommended they still get the vaccine.
“So people who get a single-dose who haven’t been infected their immune response to the vaccine is a little bit less, so it looks like having the virus actually enhances the immune response associated with the vaccine,” he said.
Venditto said both doses are recommended to reach full immunity.
Dr. Greenberg said vaccines don’t prevent someone from getting a virus, they keep people alive and prevent hospitalization.

“But it’s not a shield against any pathogen, it’s the immune response is there. And when the person becomes infected with the pathogen, immune response kicks in,” Greenberg said. “And the more active the immune response, the better chance for the person to get over the infection. They may be asymptomatic or have mild disease.”
In the Johnson & Johnson trials, the vaccine has been 100% effective in preventing hospitalizations and death from COVID-19 illness.
“Nobody died from COVID, who received the vaccine,” Greenberg said. “And in the placebo arm, there were five deaths attributed to COVID.”
In the Pfizer and Moderna trials, Venditto said, almost all symptomatic cases occurred in the placebo, or control group. “So from that we can say that the vaccines are 95% effective in reducing symptomatic infection.”
A number of concerns have been raised about the effectiveness of vaccines against COVID-19 variants. Still all three vaccines work to prevent severe illness.
“So where there were variants, it seemed to be less effective against them for mild and moderate cases,” Greenberg said of the Johnson & Johnson vaccine. “But overall, the numbers for severe cases, hospitalization and death, remain quite impressive.”
Venditto said the Moderna and Pfizer vaccines work against variants. But if the vaccine wasn’t effective against a variant, adjustments to vaccines could be made.

“The nice thing about the vaccine is the way the Pfizer, Moderna vaccines are made, if we find a variant that’s not targeted by the vaccine at all, it’s a relatively quick turnaround for Pfizer, Moderna vaccines to change that little piece of the genetic code in the vaccine,” he said.
A January study published by The New England Journal of Medicine suggested that the vaccines’ beneficial effects could be short-lived in seniors — potentially less than a year.
Other research on long-term efficacy is underway and both Greenberg and Venditto said it’s too soon to know if COVID-19 will require an annual booster.
Safety
Q: Does the vaccine have a sulfa drug in it? What if a person is allergic to sulfa drugs?
Venditto said, “I’ve not seen anything about contraindications of the Pfizer, Moderna vaccine in patients with sulfa allergies.” Anaphylactic reactions to the COVID-19 vaccines are rare, he said. Out of every million doses administered, depending on the vaccine, two to five anaphylactic reactions have occurred. However, he recommended that if you carry an EpiPen you should bring it with you when you get vaccinated.
“This is happening in such a small percentage of the population, and there’s no indication that it’s connected to sulfa drug allergies,” Venditto said. “But you should be aware that if you have an allergy, to make sure that you’re taking appropriate precautions, it’s still recommended that people with sulfa allergies or any other allergies still get immunized.”
For questions pertaining to immunosuppressant drugs or anyone who is immunocompromised, both Greenberg and Venditto advised people to discuss the vaccine with their doctors and did not make any other recommendations.
Q: I have heard you should not take Tylenol before the vaccine as it can cause a less efficient shot. Is this true?
Venditto said this is true and people should not take Tylenol (acetaminophen) or any other type of pain relievers just before getting the vaccine because they affect the immune system.
“If you take them before getting the vaccine, you can limit the immune response and you want to make sure that the vaccine has as best a chance as possible to induce that really strong protective immunity,” he said.
Tylenol or ibuprofen can be taken after the second dose of the vaccine.
“I took Tylenol after my second shot. But it wasn’t until about 12 to 18 hours after the vaccine when I was still in some discomfort,” Venditto said.
Q: What about our babies and children?
Greenberg said the University of Kentucky plans to include children from six months in age to 18 years old in future studies.
Venditto doesn’t know the status of studies that include people under the age of 16.
“All of the companies that are developing vaccines are interested in that question. But they had to show efficacy and safety in adults first before they went to children,” he said.
Eligibility and Access
The ReSource received a number of questions about who is currently eligible for the vaccine. In many cases, the answers vary from state to state because officials have set somewhat different priorities for earliest access. Kentucky, for example, made teachers and school staff eligible for early access, while some other states did not. Kentucky also recently elevated the priority for childcare workers, who were moved from group 1C to group 1B and can now receive the vaccine.
NPR created this tool to help people find when and where they can get the vaccine according to rules of the state where they live. The tool also includes links for anyone needing help signing up for a vaccine.
Q: I am 73 years old. How/when will I be able to get vaccinated? Where?
Most states are prioritizing vaccines for senior citizens, but the eligible ages vary. In West Virginia, anyone over the age of 16 can pre register for the vaccine and it’s now available to anyone 65 and older. Ohioans who are 65 and over are also eligible. Kentucky is vaccinating people over age 70.
Q: My elder sister is homebound. How will she get access to the vaccine?
Transportation can be a major hurdle, especially for those with limited mobility.
Kentucky Gov. Andy Beshear recently addressed this issue. He announced that Blue Grass Community Action Partnership is offering free rides to vaccination sites at the Kentucky Horse Park and Ephraim McDowell Hospital. To make an appointment, call 24 hours prior to your vaccination appointment. But he acknowledged that more action is needed for people who cannot travel.
“We are working on mobile units to reach those individuals maybe even in their homes themselves. The Johnson & Johnson vaccine is going to make that certainly more possible because you only have to reach them once with a single-dose vaccine,” Beshear said.
In Ohio, Medicaid recipients can call providers for transportation help. Some public and rural ride services also offer transportation to vaccine sites. All 55 West Virginia counties have vaccination sites, but traveling to the nearest site can be burdensome.
Vaccine shipments have recently been delayed by winter storms. Prior to that supplies have been limited.
Regardless of immigration status, anyone in the U.S. is eligible for the vaccine.

How It Works
Q: What does the vaccine consist of? Does it consist of a stream of the covid, like the flu shots that are given?
Typically, a vaccine includes a weakened germ to stimulate the body’s immune response, according to the Centers for Disease Control and Prevention. But COVID-19 vaccines are messenger RNA vaccines.
“When the mRNA is delivered into cells, it’s delivered in these oil droplets that help it get into the cells,” Venditto explained. “And then the mRNA is used to make the protein pieces from the virus.”
The protein pieces aren’t infectious, he said. “The process that it takes to make those proteins and then the immune system needs to respond to these protein pieces to mount that immune response.”
NOTE: This story was modified on Feb. 27 to update the status of the approval of the Johnson & Johnson vaccine and to clarify Vince Venditto’s area of expertise.
The Ohio Valley ReSource gets support from the Corporation for Public Broadcasting and our partner stations.






